Contract Manufacturing
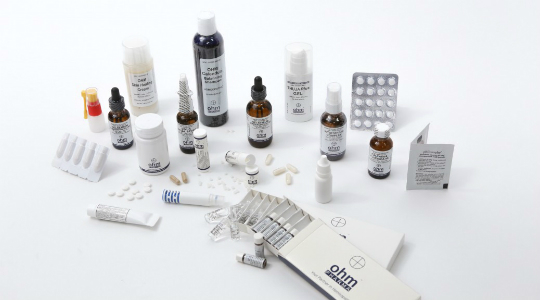
– We provide our lab, experience and knowledge for your custom projects.
– We manufacture single remedies in X, C and LM potencies, kits: 12, 25, 50 or 100 remedies, spagyric tinctures, combination remedies, homacords, lactose free pellets, tablets, capsules, powders, syrups, nasal sprayers, ampoules, suppositories (vaginal or rectal), shampoos, topical sprayers, gels, creams, ointments and other pharmaceutical forms.
– We assure regulatory compliance with cGMP, FDA, DOH and HPUS guidelines and regulations.
– We hold OTC and Rx drug manufacturing licenses.
– OHM PHARMA is registered with FDA and Health Canada
– We assist in FDA company and product registration (NDC codes).
– When you partner with us we can assure you with certainty that the outcome will exceed your expectations.




